 :max_bytes(150000):strip_icc()/lithiumatom-56a12c335f9b58b7d0bcc103.jpg)
The general layout here was taken from Rohlf but some discrepancies were found in the lower levels. This is true for high angular momentum states as shown, but the s and p states fall well below the corresponding hydrogen energy levels. Since the outer electron looks inward at just one net positive charge, it could be expected to have energy levels close to those of hydrogen. The lithium atom has a closed n=1 shell with two electrons and then one electron outside. The lithium model can be used with fluoride to demonstrate that an ionic compound such as salt is more than an organized structure made up of plain, solid spheres.Hydrogen-Like Atoms:Lithium Lithium Energy Levels Develop models to describe the atomic composition of simple molecules and extended structure. Parents have reported that some of the students have kept and displayed their model for as many as ten years after graduation. 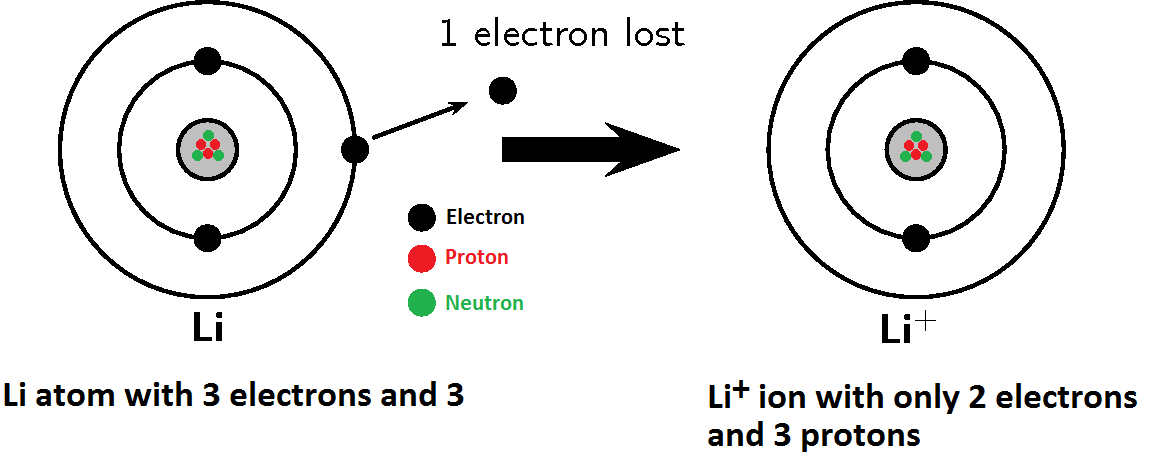
Some teachers have already reported to us that the models have stimulated conversations among students relevant to the proposed lesson before the teacher even begins to interact with the students.Ĭompletion of the physical model has been reported to give many students a sense of pride in being able to do more than read and respond. A lesson that sometimes is just more fun.A model that is a step or two closer to reality than printed text or drawings.Hands-on learning environment that develops critical thinking.A great hands-on manipulative for classroom activities that can engage the student in active learning with more material retained.Also useful for: PS1.A: Structure and Properties of MatterĪs noted already, the model provides a three dimensional display of the structure of the lithium atom which is relevant to NGSS standard PS1.A: Structure and Properties of Matter and provides the following: This lithium atom model can be used with other atom models from periods 1, 2, 3, and 4 to analyze periodic trends. (Hint: introduce a fluorine atom into the lesson! and discuss formula units and/or molecules.) The challenge will be for students to solve the neutrality problem and still keep lithium stable. With only two electrons the lithium may be stable, but it is no longer neutral.The empty second energy level orbitals can be removed leaving a lithium with two electrons in the first energy level, now the outermost energy level. If the one valence electron, which can be removed from the hole, is taken out, the entire second energy level is left with eight empty holes. Thus lithium is not stable as presented by this model. Lithium has one valence electron in its outermost energy level and seven empty holes that could hold electrons.There are lessons available specifically about isotopes! that deal with variation neutron numbers.) (Since neutron numbers vary they are not shown in these models. The lithium model displays three protons in the nucleus and three electrons in the first and second energy levels.This means two electrons for hydrogen and helium and eight electrons for all other atoms (octet rule). Stability is accomplished when the outermost energy level is filled with electrons, or totally empty. Neutrality is accomplished with number of electrons being equal to number of protons. Quantum mechanics requires that all atoms are both neutral and stable.The electrons in the model are not real, but they are a step closer to reality than written symbols and help to understand what the arrows and other symbols in orbital diagrams and electron configurations represent. The symbol for an electron, core or valence, in an orbital diagram is an arrow. This model can be dismantled so that the orbitals can be placed on an orbital diagram which is available from Quanta to Galaxies.Valence electrons can go, or leave the atom as indicated by green.) 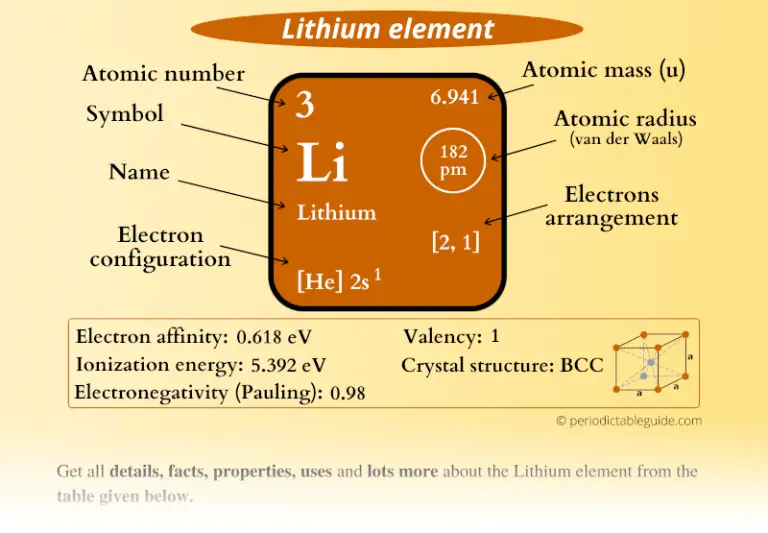
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
